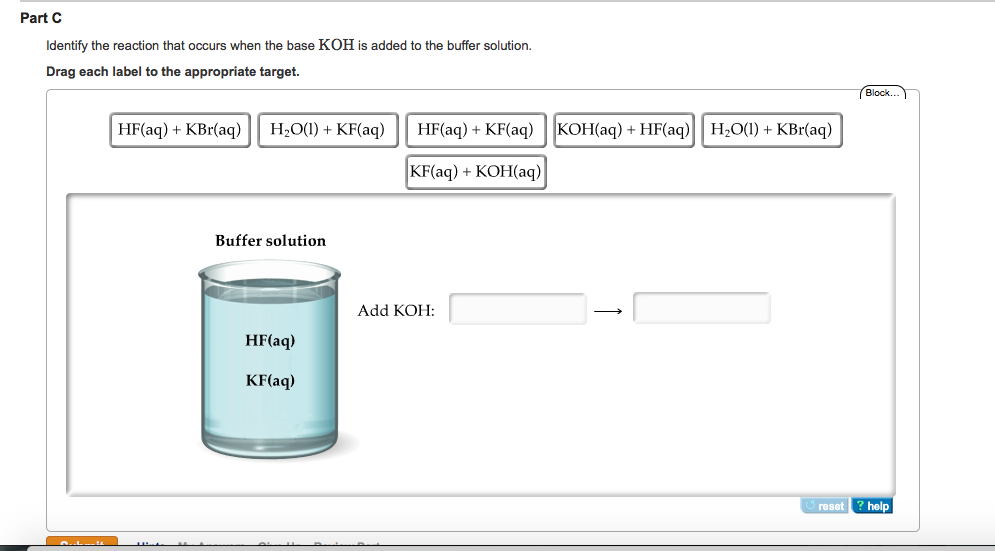
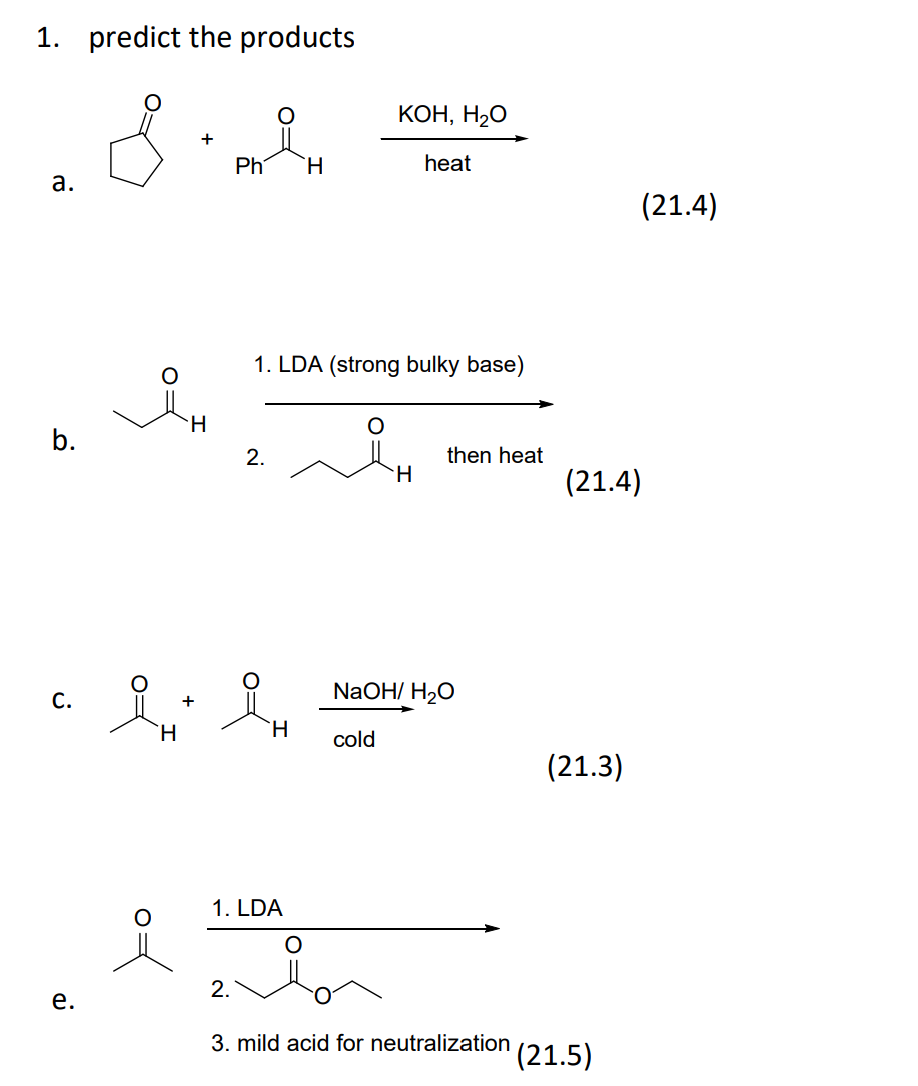
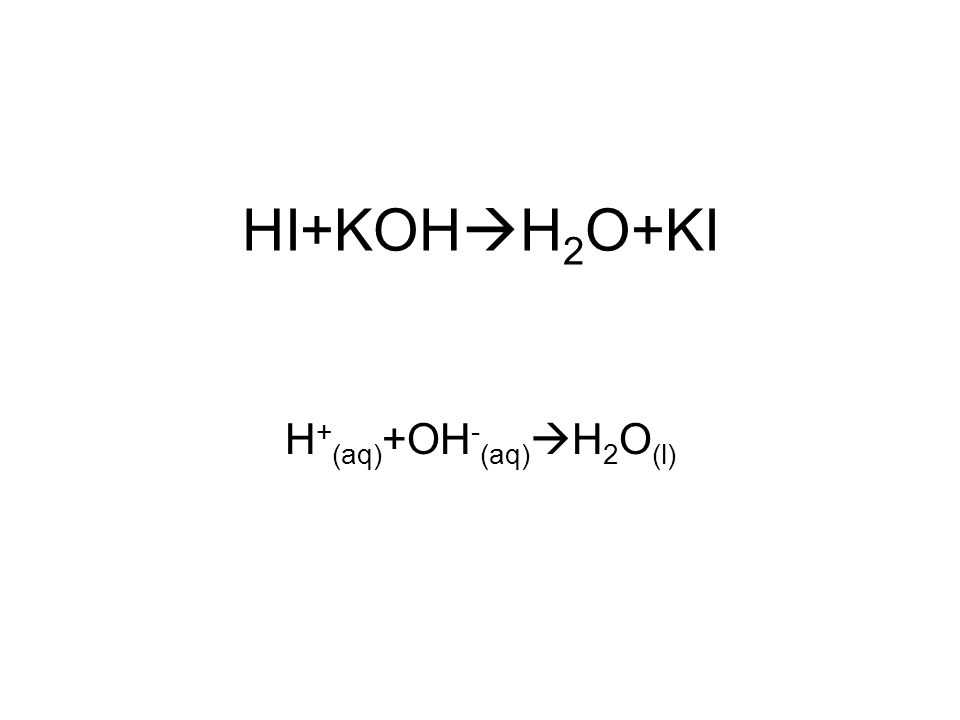
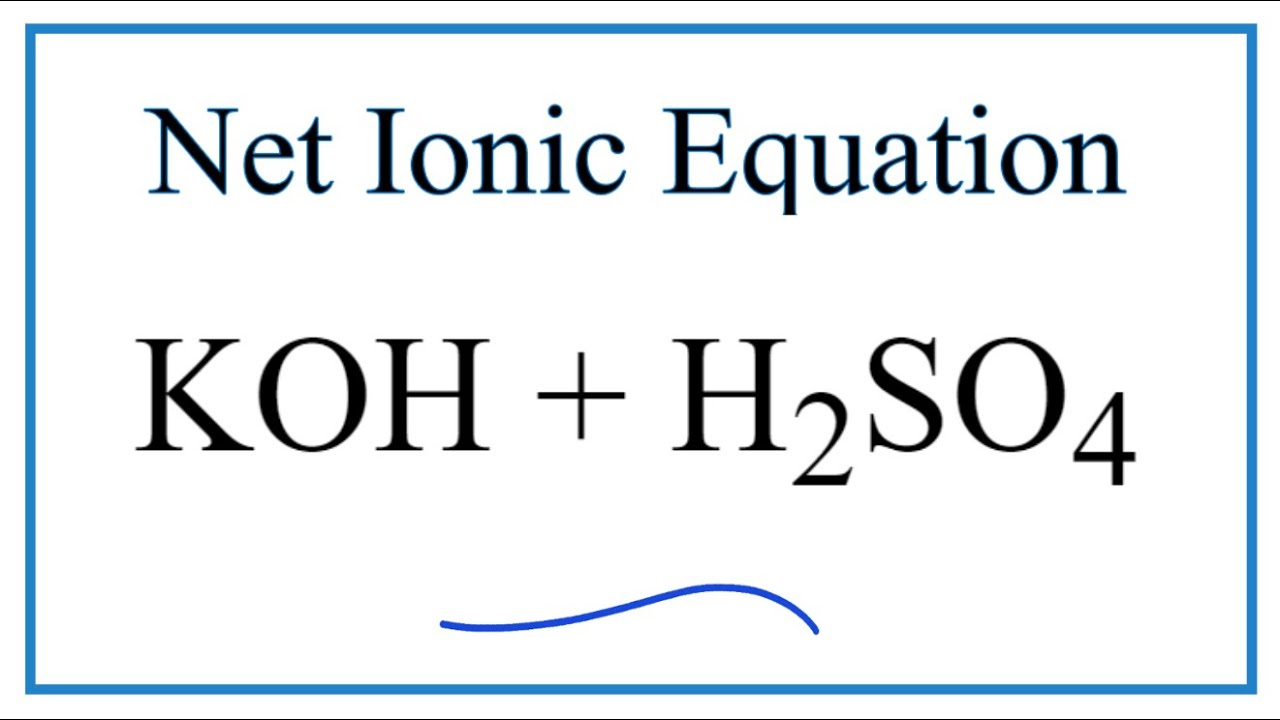SOLVED: The balanced chemical equation for the reaction of the base potassium hydroxide with phosphoric acid to form potassium phosphate and water is: 3 KOH HPO4 KaPOs 3 HzO How many grams

Selective Focus of a Bottle of Pure Potassium Hydroxide or KOH Chemical Compound beside a Petri Dish with White Solid Pellets. Stock Photo - Image of flakes, base: 199192488
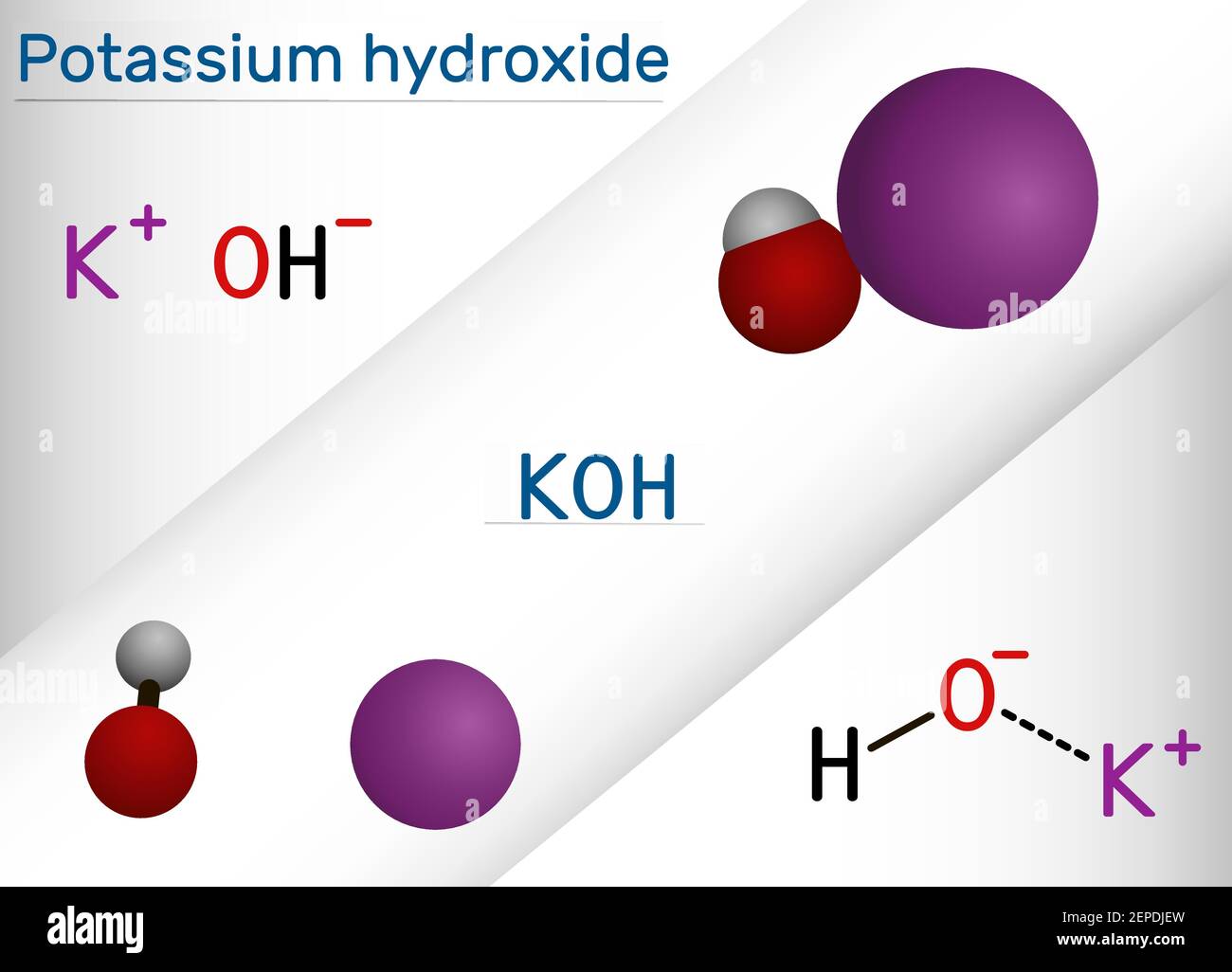
Potassium hydroxide, caustic potash, lye molecule. KOH is strong caustic base and alkali, ionic compound. Structural chemical formula and molecule mod Stock Vector Image & Art - Alamy