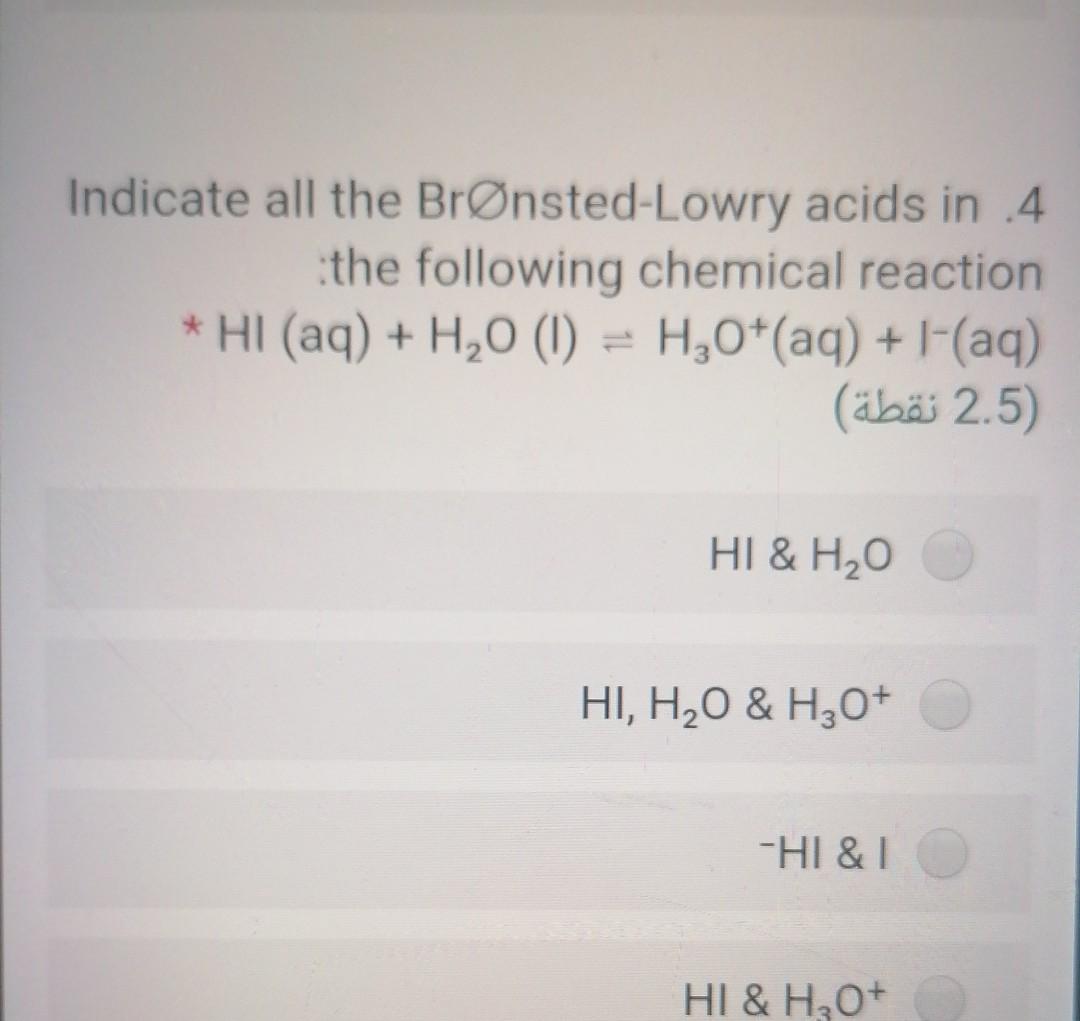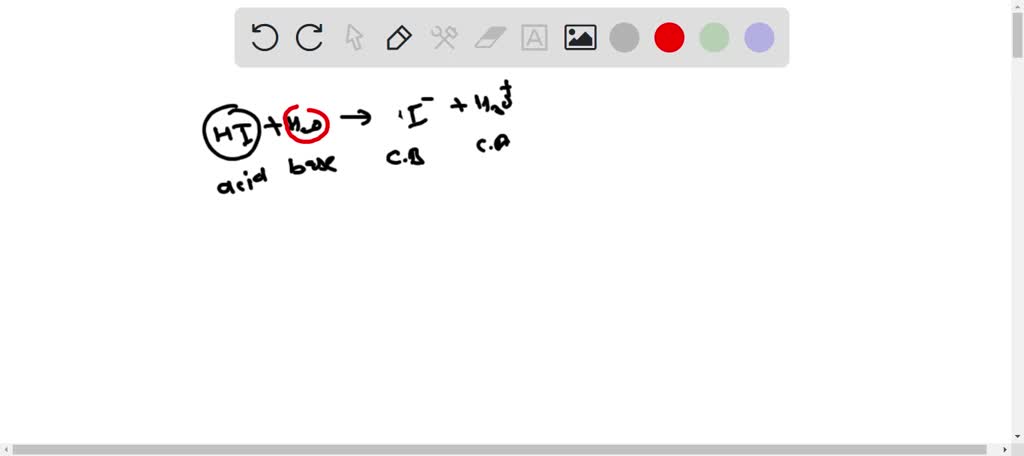
SOLVED: Hydroiodic acid is a strong acid that reacts with water as shown below: HI(aq) + H2O(aq) –> I- (aq) + H3O+ (aq) a. Identify the acid, base, conjugate acid, and conjugate
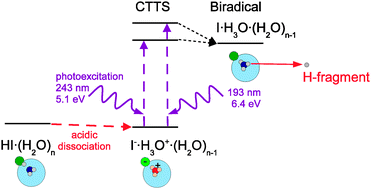
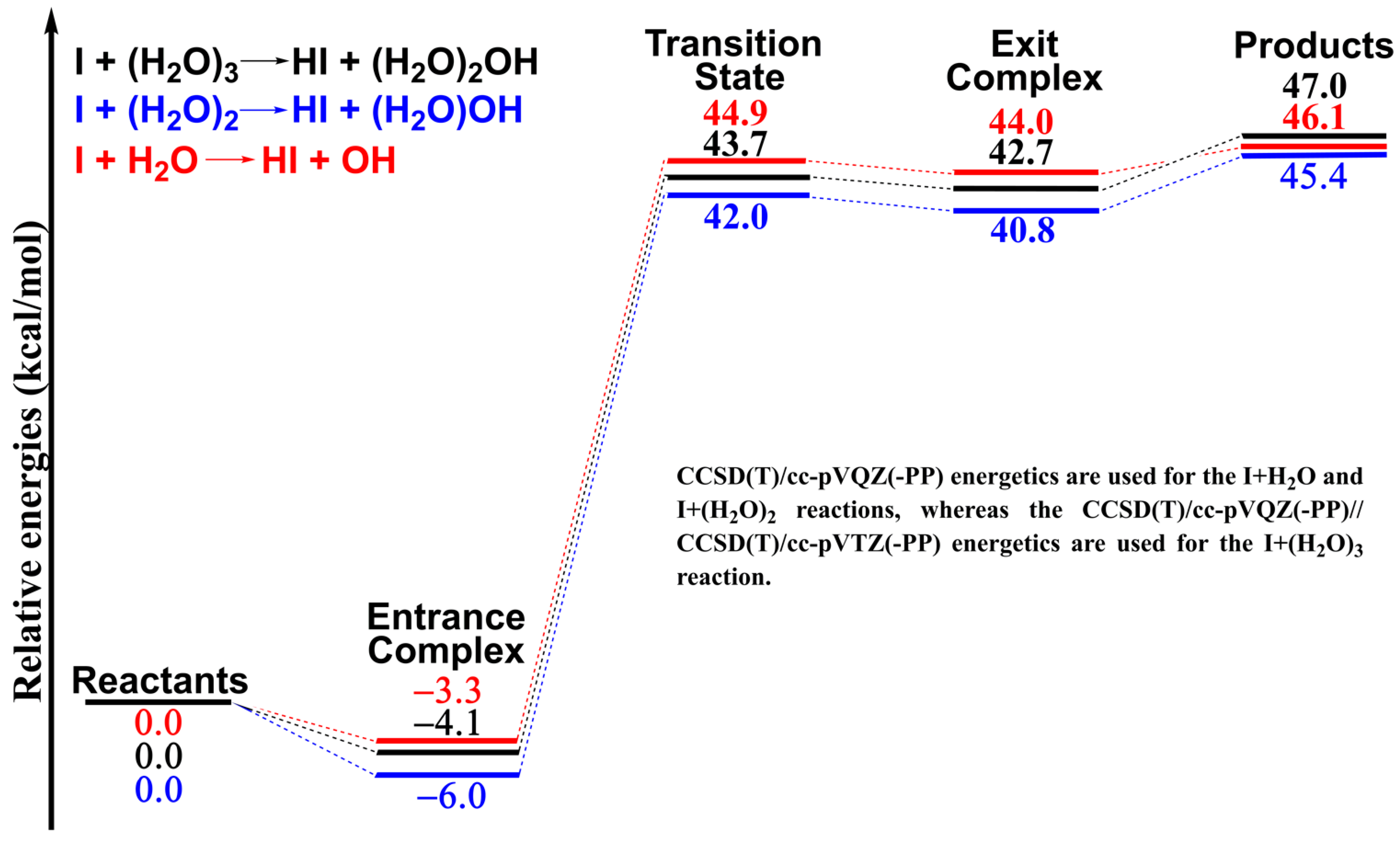
Photochemistry of HI on argon and water nanoparticles: Hydronium radical generation in HI·(H2O)n - Physical Chemistry Chemical Physics (RSC Publishing)