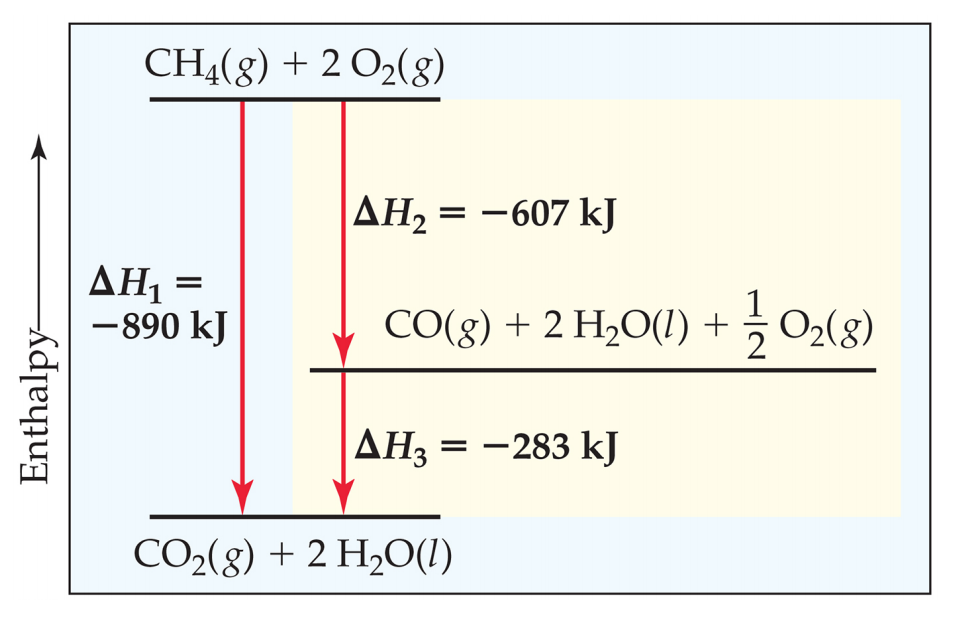
SOLVED: Given the chemical equation Na(s) + H2O(ℓ) → NaOH(aq) + H2(g) Balance the equation. How many molecules of H2 are produced when 332 atoms of Na react?
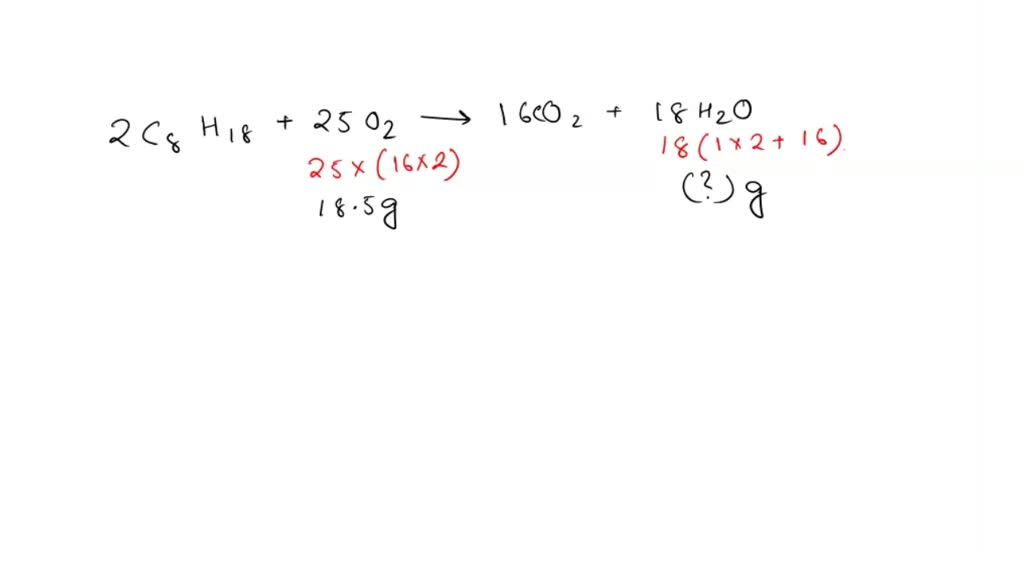
SOLVED: According to the balanced chemical equation below, how many grams of H2O would be produced if 18.5 grams of O2 reacted with excess C8H18? 2 C8H18 + 25 O2 –> 16 CO2 + 18 H2O