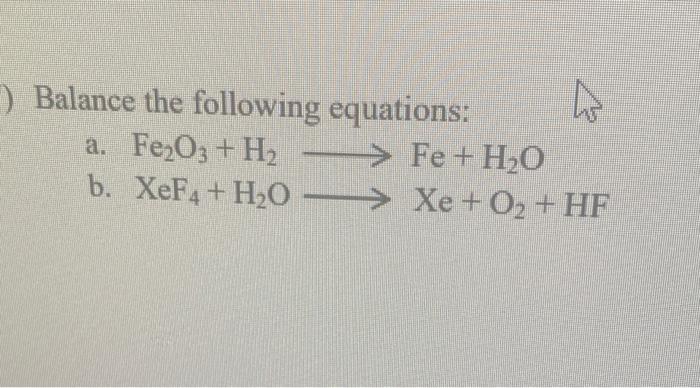SOLVED: Using the following equation: Fe2O3 + H2 –> Fe + H2O Calculate how many grams of iron (Fe) can be made from 16.5 grams of Fe2O3 from the chemical equation. PLEASE SHOW WORK!!!
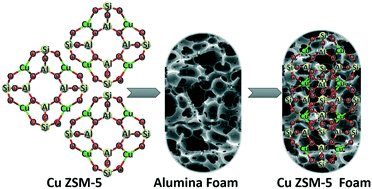
Selective catalytic reduction of NO over hierarchical Cu ZSM-5 coated on an alumina foam support - Reaction Chemistry & Engineering (RSC Publishing)
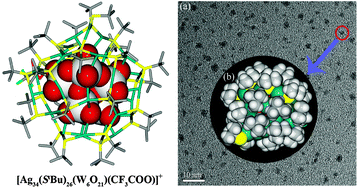
Assembly of a luminescent core–shell nanocluster featuring a Ag34S26 shell and a W6O216− polyoxoanion core - Chemical Communications (RSC Publishing)
![SOLVED: Determine the equilibrium constant for the reaction between iron (III) oxide and hydrogen. Fe2O3 (s) + 3H2 (g) 2Fe (s) + 3H2O (g) Group of answer choices Kc = [H2O]3 / [ SOLVED: Determine the equilibrium constant for the reaction between iron (III) oxide and hydrogen. Fe2O3 (s) + 3H2 (g) 2Fe (s) + 3H2O (g) Group of answer choices Kc = [H2O]3 / [](https://cdn.numerade.com/ask_previews/d4d6e0e8-f43e-406e-85e1-9586a85b0f23_large.jpg)