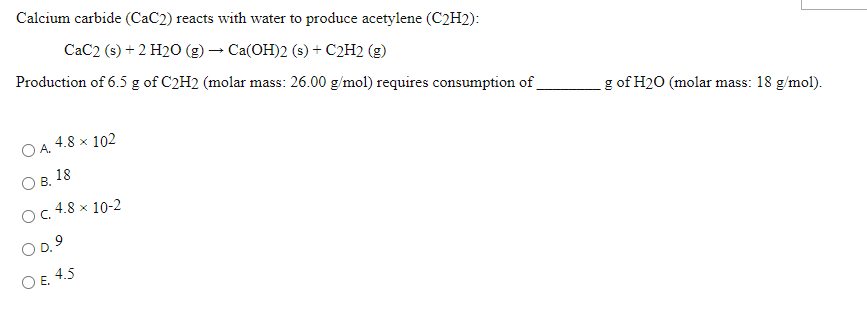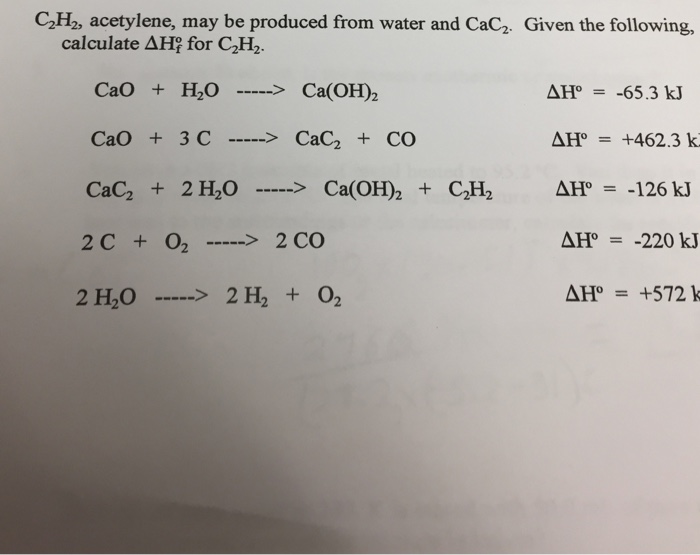Chemical Equations And balancing equations. Chemical Equation CH 4 + O 2 CO 2 + H 2 O Reactantsproducts Means to produce. - ppt download

How to Balance CaC2+H2O=Ca(OH)2+C2H2|Chemical equation CaC2+H2O =Ca(OH)2+C2H2|CaC2+H2O=Ca(OH)2+C2H2 - YouTube
CaC2 reacts with H2O and gives X. X reacts with Cu2Cl2 in the presence of NH4OH and give Y. What is Y, and how? - Quora
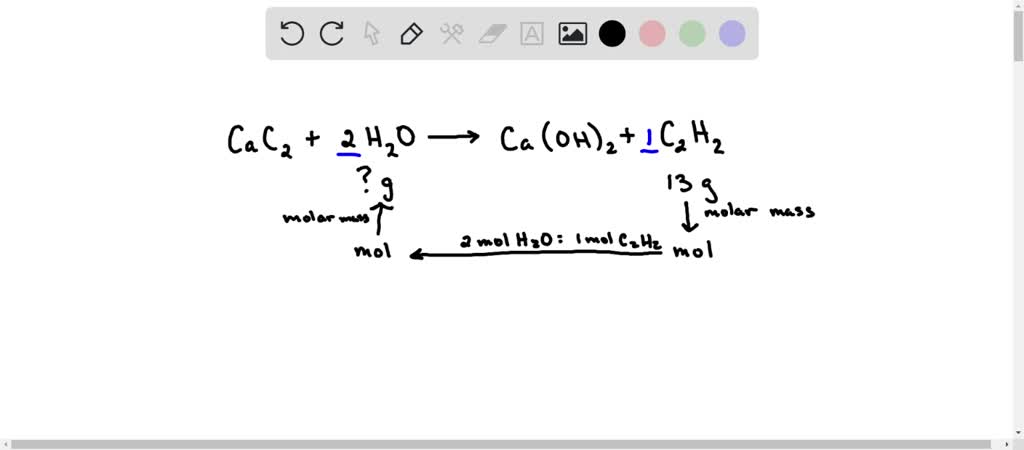
SOLVED: Calcium carbide (CaC2) reacts with water to produce acetylene (C2H2): CaC2 (s) + 2H2O (g) → Ca(OH)2 (s) + C2H2 (g) Production of 13 g of C2H2 (26.04 g/mol) requires consumption

Class11Cac2+H2O=acetylene (calcium carbide react with water)complete reaction explanations in Telugu - YouTube