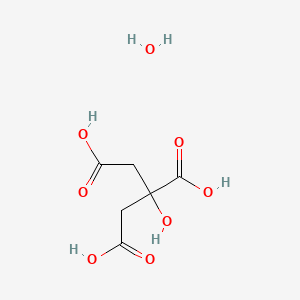
Citric acid (C6H8O7) is 7.55% dissociated in a solution prepared by dissolving 0.240 mole of acid in two liters of solution. How would I find the Ka of this? - Quora

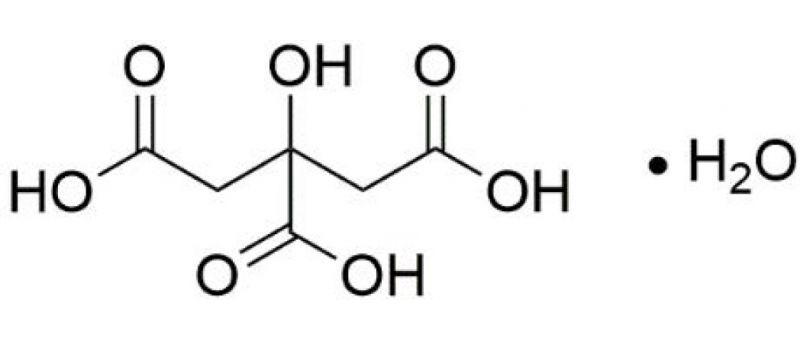
CITRIC ACID (monohydrate) Extra Pure Assay : Min. 99% C6H8O7.H2O M.W. 210.14 500GM : Amazon.in: Industrial & Scientific
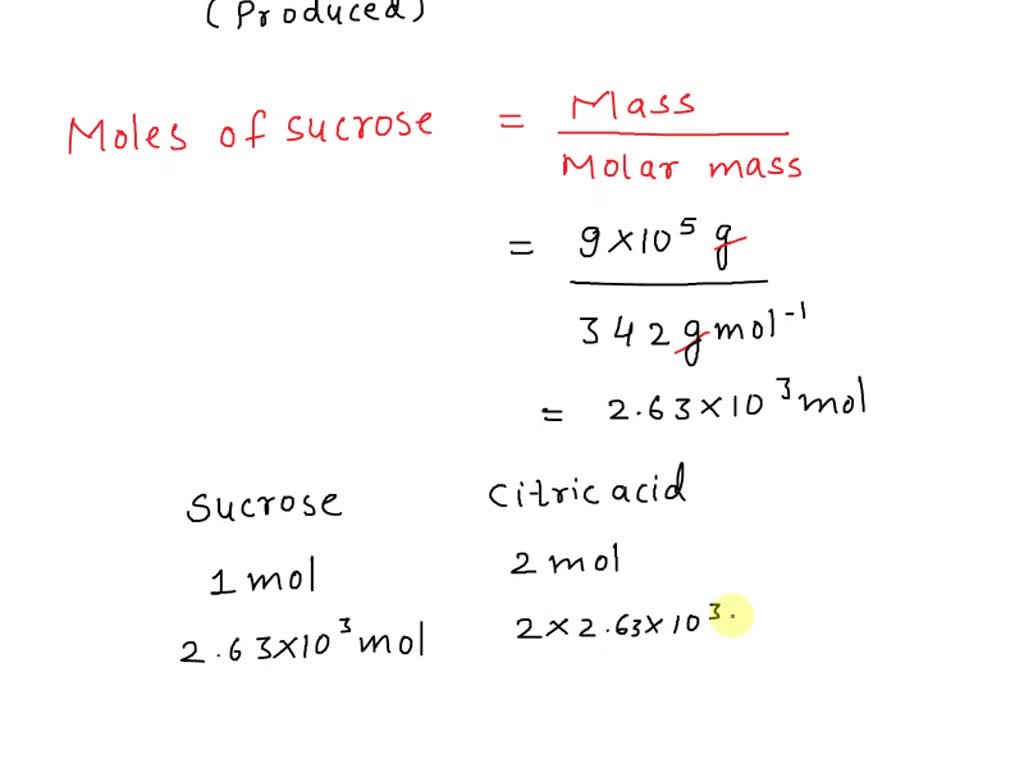
SOLVED: Citric acid ce(C6H8O7)(CX6HX8OX7) , named for its natural occurrence in citrus fruits, is produced in large quantities industrially. This is often achieved through large-scale fermentation of sucrose and/or glucose. The reaction
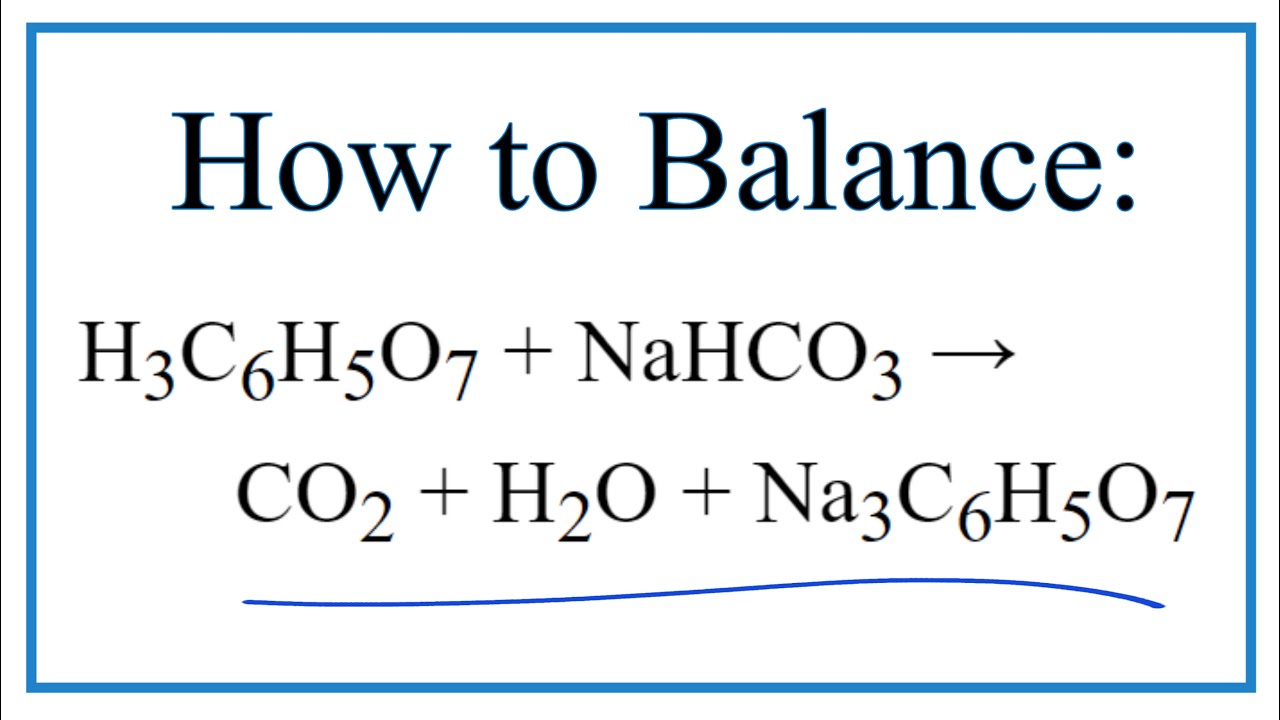
How to Balance H3C6H5O7 + NaHCO3 = CO2 + H2O + Na3C6H5O7 (Citric acid + Sodium bicarbonate ) - YouTube

4.66 | Citric acid, C6H8O7, a component of jams, jellies, and fruity soft drinks, is prepared - YouTube