Optimized Cluster Structures of trans-Carboxyl (1Ht) with 6 to 11 H2O... | Download Scientific Diagram

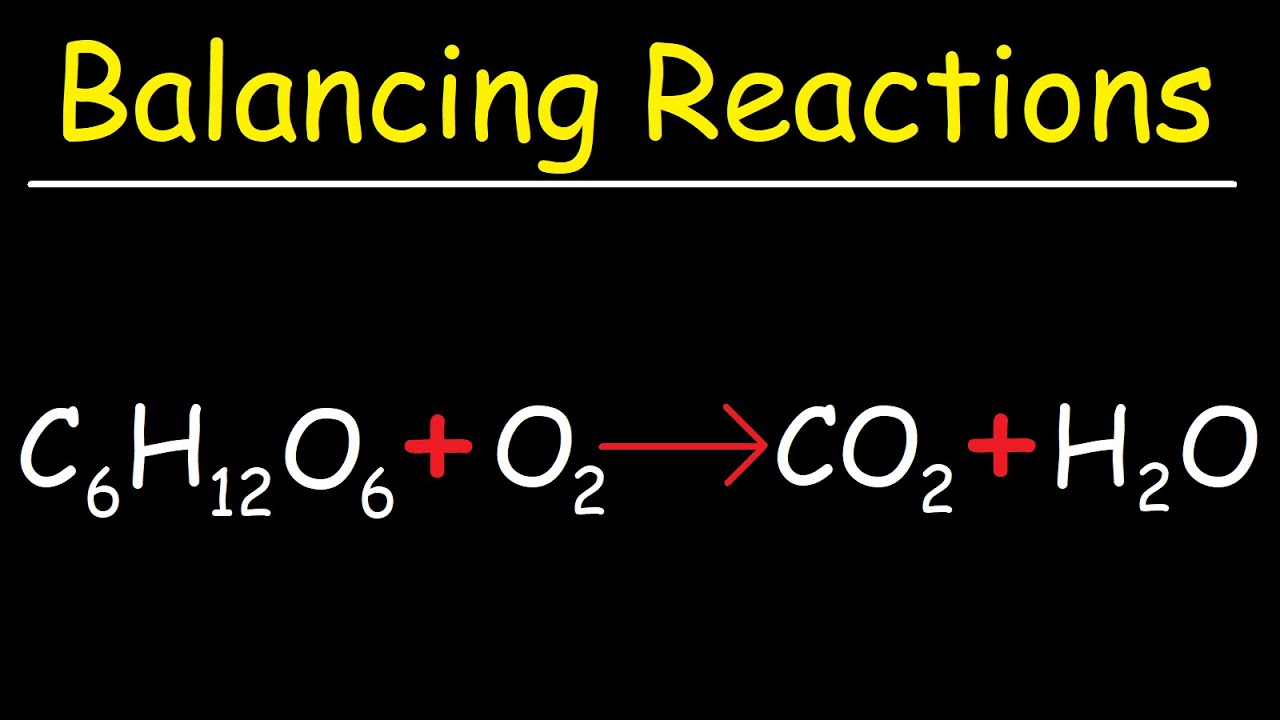
SOLVED: C6H12O6 + 6 O2 –> 6 CO2 + 6 H2O After burning your marshmallows, you capture 36.0 g of water. How many Liters of H2O did your combustion reaction produce?
FeCl3·6H2O catalyzed aqueous media domino synthesis of 5-monoalkylbarbiturates: water as both reactant and solvent - RSC Advances (RSC Publishing)
![SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [ SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [](https://cdn.numerade.com/ask_previews/e7bf0f40-8942-4c62-a0e8-90258b6ac48b_large.jpg)
SOLVED: NiCl2·6 H2O + 3 C2H8N2 → [Ni(C2H8N2)3]Cl2 + 6 H2O Nick, Ethyl, and Amy reacted 3.60 g of NiCl2·6 H2O with 8.50 mL of 4.0 M C2H8N2and obtained 2.83 g of [

CaCl2.6H2O(s) CaCl2(s) + 6H2O(g); Kp = 6.4 × 10^-17 . Excess solid CaCl2. 6H2O and CaCl2 are taken in a container containing some water vapours at a pressure of 1.14 torr at a
![SOLVED: Cobalt (II) Ion, Water, and Chloride Ion [Co(H2O)6]2+ + 4 Cl- ⇄ [CoCl4]2- + 6 H2O Pink Blue Addition of 12M HCl to the Co(NO3)2 solution: Addition of water: Addition of SOLVED: Cobalt (II) Ion, Water, and Chloride Ion [Co(H2O)6]2+ + 4 Cl- ⇄ [CoCl4]2- + 6 H2O Pink Blue Addition of 12M HCl to the Co(NO3)2 solution: Addition of water: Addition of](https://cdn.numerade.com/ask_previews/955ea8da-de3b-4037-b192-98361234d9a3_large.jpg)
SOLVED: Cobalt (II) Ion, Water, and Chloride Ion [Co(H2O)6]2+ + 4 Cl- ⇄ [CoCl4]2- + 6 H2O Pink Blue Addition of 12M HCl to the Co(NO3)2 solution: Addition of water: Addition of
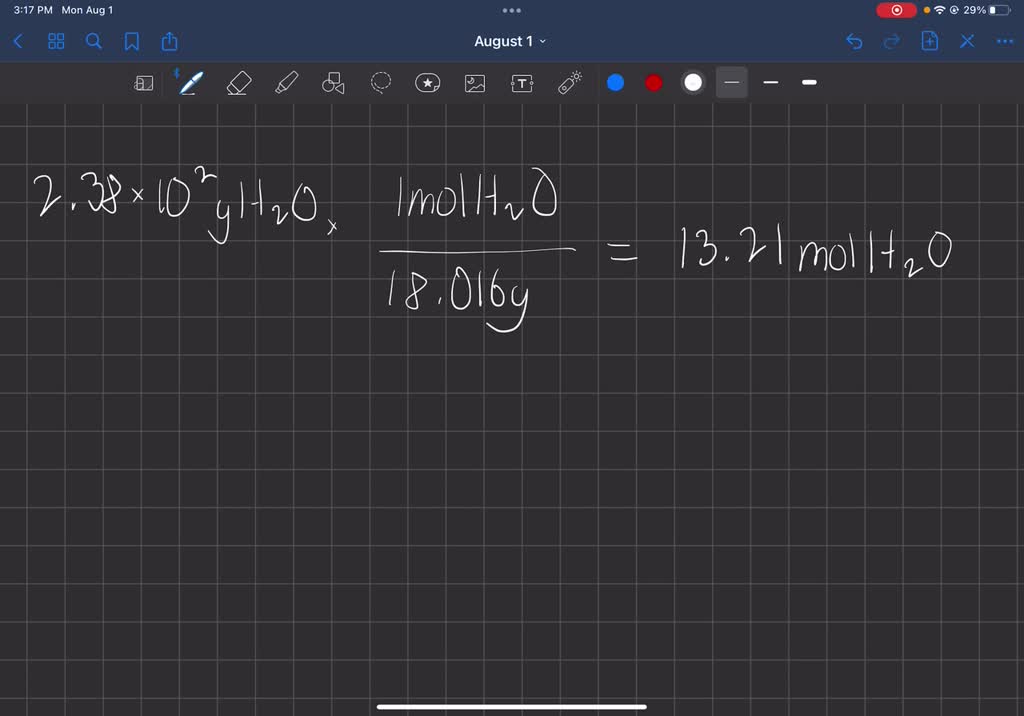
SOLVED: Consider this reaction: 6 CO2 + 6 H2O + light C6H12O6 + 6 O2If there were 2.38 x 102 g of H2O, 18.6 moles of CO2, and plenty of light, what would be the theoretical yield of the reaction?